Abstract
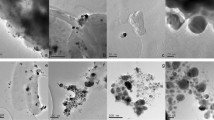
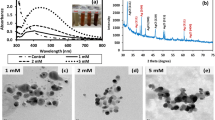
Silver nanoparticles (AgNPs), embedded into a specific exopolysaccharide (EPS), were produced by Klebsiella oxytoca DSM 29614 by adding AgNO3 to the cultures during exponential growth phase. In particular, under aerobic or anaerobic conditions, two types of silver nanoparticles, named AgNPs-EPSaer and the AgNPs-EPSanaer, were produced respectively. The effects on bacterial cells was demonstrated by using Escherichia coli K12 and Kocuria rhizophila ATCC 9341 (ex Micrococcus luteus) as Gram-negative and Gram-positive tester strains, respectively. The best antimicrobial activity was observed for AgNPs-EPSaer, in terms of minimum inhibitory concentrations and minimum bactericidal concentrations. Observations by transmission electron microscopy showed that the cell morphology of both tester strains changed during the exposition to AgNPs-EPSaer. In particular, an electron-dense wrapped filament was observed in E. coli cytoplasm after 3 h of AgNPs-EPSaer exposition, apparently due to silver accumulation in DNA, and both E. coli and K. rhizophila cells were lysed after 18 h of exposure to AgNPs-EPSaer. The DNA breakage in E. coli cells was confirmed by the comparison of 3-D fluorescence spectra fingerprints of DNA. Finally the accumulation of silver on DNA of E. coli was confirmed directly by a significant Ag+ release from DNA, using the scanning electrochemical microscopy and the voltammetric determinations.





Similar content being viewed by others
References
Angelova T, Rangelova N, Dineva H, Georgieva N, Müller R (2014) Synthesis, characterization and antibacterial assessment of SiO2-hydroxypropylmethyl cellulose hybrid material with embedded silver nanoparticles. Biotechnol Biotechnol Equip 28:747–752
Arcon I, Paganelli S, Piccolo A, Gallo M, Vogel-Mikus K, Baldi F (2015) XAS analysis of iron and palladium bonded to a polysaccharide produced anaerobically by a strain of Klebsiella oxytoca. J Synchrotron Radiat 22:1215–1226
Baldi F, Minacci A, Pepi M, Scozzafava A (2001) Gel sequestration of heavy metals by Klebsiella oxytoca isolated from iron mat. FEMS Microbiol Ecol 36:169–174
Baldi F, Marchetto D, Zanchettin D, Sartorato E, Paganelli S, Piccolo O (2010) A bio-generated Fe(III)-binding exopolysaccharide used as new catalyst for phenol hydroxylation. Green Chem 12:1405–1409
Baldi F, Marchetto D, Paganelli S, Piccolo O (2011) Bio-generated metal binding polysaccharides as catalysts for synthetic applications and organic pollutant transformation. New Biotechnol 29:74–78
Battistel D, Baldi F, Gallo M, Faleri C, Daniele S (2015) Characterisation of biosynthesised silver nanoparticles by scanning electrochemical microscopy (SECM) and voltammetry. Talanta 132:294–300
Dayem AA, Kim BW, Gurunathan S, Choi HY, Yang G, Saha SK et al (2014) Biologically synthesized silver nanoparticles induce neuronal differentiation of SH-SY%Y cells via modulation of reactive oxygen species, phosphatases, and kinase signaling pathways. Biotechnol J 9:934–943
Deepak V, Kalishwaralal K, Pandian SRK, Gurunathan S (2011) An insight into bacterial biogenesis of silver nanoparticles, industrial production and scale-up. In: Duran MRN (ed) Metal nanoparticles in microbiology. Springer, Berlin, pp 17–35
Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO (2000) A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J Biomed Mater Res A 52:662–668
Gallo G, Baldi F, Renzone G, Gallo M, Cordaro A, Scaloni A, Puglia AM (2012) Adaptative biochemical pathways and regulatory networks in Klebsiella oxytoca BAS-10 producing a biotechnologically relevant exopolysaccharide during Fe(III)-citrate fermentation. Microb Cell Factories 11:152–168
George L, Sankaran K, Viswanathan KS, Mathews CK (1994) Matrix-isolation infrared spectroscopy of organic phosphates. Appl Spectrosc 48:7–12
Gwinn EG, O’Neill P, Guerrero AJ, Bouwmeester D, Fygenson DK (2008) Sequence-dependent fluorescence of DNA-hosted silver nanoclusters. Adv Mater 20:279–283
Gwinn EG, Schultz D, Copp SM, Swansey S (2015) DNA-protected silver cluster for nanophotonics. Nanomaterials 5:180–207
Hadioui M, Leclerc S, Wilkinson KJ (2013) Multimethod quantification of Ag release from nanosilver. Talanta 105:15–19
Holt KB, Bard AJ (2005) Interaction of Silver(I) ions with the respiratory chain of Escherichia coli: an electrochemical and scanning electrochemical microscopy study of the antimicrobial mechanism of micromolar Ag+. Biochemistry 44:13214–13223
Kanmani P, Lim ST (2013) Synthesis and structural characterization of silver nanoparticles using bacterial exopolysaccharide and its antimicrobial activity against food and multidrug resistant pathogens. Process Biochem 48:1099–1106
Khan SS, Mukherjee A, Chandrasekaran N (2011) Studies on interaction of colloidal silver nanoparticles (SNPs) with five different bacterial species. Colloids Surf B 87:129–138
Lee W, Kim K-H, Lee DG (2014) A novel mechanism for the antibacterial effect of silver nanoparticles on Escherichia coli. Biometals 27:1191–1201
Leone S, De Castro C, Parrilli M, Baldi F, Lanzetta R (2007) Structure of the iron binding polysaccharide produced anaerobically by the Gram-negative bacterium Klebsiella oxytoca BAS-10. Eur J Org Chem 31:5183–5189
Li WR, Xie XB, Shi QS, Zeng HY, Ou-Yang YS, Chen YB (2010) Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl Microbiol Biotechnol 85:1115–1122
Li G, He D, Qian Y, Guan B, Gao S, Cui Y et al (2012) Fungus-mediated green synthesis of silver nanoparticles using Aspergillus terreus. Int J Mol Sci 13:446–476
McQuillan JS, Infante HG, Stokes E, Shaw AM (2012) Silver nanoparticle enhanced silver ion stress response in Escherichia coli K12. Nanotoxicology 6:857–866
Mijnendonckx K, Leys N, Mahillon J, Silver S, van Houdt R (2013) Antimicrobial silver: uses, toxicity and potential for resistance. Biometals 26:609–621
Narayanan KB, Sakthivel N (2010) Biological synthesis of metal nanoparticles by microbes. Adv Colloid Interface Sci 156:1–13
Okafor F, Janen A, Kukhtareva T, Edwards V, Curley M (2013) Green synthesis of silver nanoparticles, their characterization, application and antibacterial activity. Int J Environ Res Public Health 10:5221–5238
Paganelli S, Tassini R, La Sorella G, Piccolo O, Baldi F, Rathod VD (2015) Aqueous biphasic treatment of some nitrocompounds with hydrogen in the presence of a biogenerated Pd-polysaccharide. New Biotechnol 32:313–317
Pal S, Tak YK, Song JM (2007) Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli. Appl Environ Microbiol 73:1712–1720
Panacek A, Kvitek L, Prucek R, Kolar M, Vecerova R, Pizurova NJ (2006) Silver colloid nanoparticles: synthesis, characterization, and their antibacterial activity. J Phys Chem 110:16248–16253
Paredes D, Ortiz C, Torres R (2014) Synthesis, characterization, and evaluation of antibacterial effect of Ag nanoparticles against Escherichia coli O157:H7 and methicillin-resistant Staphylococcus aureus (MRSA). Int J Nanomed 9:1717–1729
Pospiech A, Neumann B (1995) A versatile quick-prep of genomic DNA from gram-positive bacteria. Trends Genet 11:217–218
Prakash P, Gnanaprakasam P, Emmanuel R, Arokiyaraj S, Saravanan M (2013) Green synthesis of silver nanoparticles from leaf extract of Mimusops elengi, Linn. for enhanced antibacterial activity against multi drug resistant clinical isolates. Colloids Surf B 108:255–259
Qiang L, Yumei L, Sheng H, Yingzi L, Dongxue S, Dake H et al (2013) Optimization of fermentation conditions and properties of an Exopolysaccharide from Klebsiella sp. H-207 and application in adsorption of hexavalent chromium. PLoS One 8:e53542
Rai MK, Deshmukh SD, Ingle AP, Gade AK (2012) Silver nanoparticles: the powerful nanoweapon against multi-drug-resistant bacteria. J Appl Microbiol 112:841–852
Rigo C, Zamego L, Rampazzo G, Argese E (2009) Characterization of a former dump site in the Lagoon of Venice contaminated by municipal solid waste incinerator bottom ash, and estimation of possible environmental risk. Chemosphere 77:510–517
Ruparelia J, Chatterjee A, Duttagupta S, Mukherji S (2008) Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater 4:707–716
Sintubin L, De Gusseme B, Van der Meeren P, Pycke BFG, Verstraete W, Nico Boon N (2011) The antibacterial activity of biogenic silver and its mode of action. Appl Microbiol Biotechnol 91:153–162
Soltzberg LJ, Lor S, Okey-Igwe N, Newman R (2012) 3D fluorescence characterization of synthetic organic dyes. Am J Anal Chem 3:622–631
Sotiriou GA, Pratsini SE (2010) Antibacterial activity of nanosilver ions and particles. Environ Sci Technol 44:5649–5654
Sugihara R, Yoshimura M, Mori M, Kanayama N, Hikida M, Ohmori H (2000) Prevention of collagen-induced arthritis in DBAr1 mice by oral administration of AZ-9, a bacterial polysaccharide from Klebsiella oxytoca. Immunopharmacology 49:325–333
Taglietti A, Diaz Fernandez YA, Amato E, Cucca L, Dacarro G, Grisoli P et al (2012) Antibacterial activity of glutathione-coated silver nanoparticles against Gram positive and Gram negative bacteria. Langmuir 28:8140–8148
Tajkarimi M, Iyer D, Tarrannum M, Cunningham Q, Sharpe I et al (2014) The effect of silver nanoparticle size and coating on Escherichia coli. JSM Nanotechnol Nanomed 2:1025–1027
Vigneshwaran N, Nachane RP, Balasubramanya RH, Varadarajan PVA (2006) Novel one-pot ‘green’ synthesis of stable silver nanoparticles using soluble starch. Carbohydr Res 341:2012–2018
Vishnupriya S, Chaudari K, Jagannathan R, Pradepp T (2013) Single-cell investigations of silver nanoparticle-bacteria interactions. Part Part Syst Charact 30:1056–1062
Wei D, Sun W, Qian W, Ye Y, Ma X (2009) The synthesis of chitosan-based silver nanoparticle sand their antibacterial activity. Carbohydr Res 344:2375–2382
Xiu ZM, Ma J, Alvarez PJ (2011) Differential effect of common ligands and molecular oxygen on antimicrobial activity of silver nanoparticles versus silver ions. Environ Sci Technol 45:9003–9008
Zeiri I, Bronk BV, Shabtai Y, Eichler J, Efrima S (2004) Surface-enhanced Raman spectroscopy as a tool for probing specific biochemical components in bacteria. Appl Spectrosc 58:33–40
Acknowledgments
Financial support of the Ministry of University and Scientific Research (MIUR) (PRIN-2010AXENJ8) and of Cà Foscari University (ADIR) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Baldi, F., Daniele, S., Gallo, M. et al. Polysaccharide-based silver nanoparticles synthesized by Klebsiella oxytoca DSM 29614 cause DNA fragmentation in E. coli cells. Biometals 29, 321–331 (2016). https://doi.org/10.1007/s10534-016-9918-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-016-9918-4